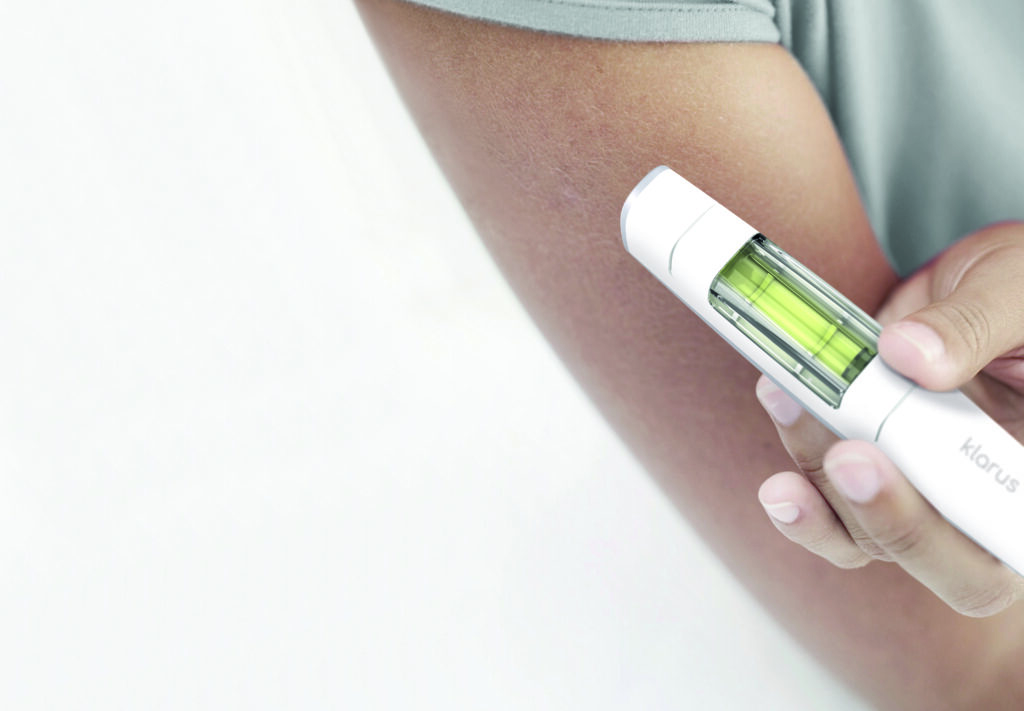
Drug Delivery
Accelerating Development and Enhancing User Experience
As the world’s healthcare needs change, innovation in drug delivery becomes increasingly vital. With rising chronic conditions and an aging population, optimizing medication delivery and enhancing patient experience are essential. Advancements will be driven by novel formulations, cutting-edge technologies, and expanding markets. To thrive, companies must address several challenges, ensuring devices meet market and user needs, adhering to stringent quality and regulatory standards, and controlling costs.
Accelerate Development
We fast-track the development of drug delivery systems from stakeholder research to clinical manufacturing.

Optimize User Experience
We enhance the user experience of drug delivery devices, ensuring they are intuitive and effective.

Specialized in Advanced Delivery Technologies
Our expertise supports established delivery routes such as parenteral (including fixed and variable dose injectors, large volume on-body delivery systems, infusion pumps), respiratory (inhalation, deep lung, nasal), sublingual, and transdermal. Additionally, we extend to novel drug delivery routes, addressing challenges such as ocular delivery, brain delivery, and other direct-to-organ delivery.
Collaborating with Re-Vana Therapeutics on a novel delivery system for the sustained release of active pharmaceutical ingredient (API) ophthalmic formulations.
From Opportunity to Clinic & Commercial
Understanding Clients’ Program Requirements
We have a deep understanding of our clients’ markets and the needs of all stakeholders. Our integrated approach ensures every decision aligns with strategic goals, balancing long-term value with immediate needs to keep clients at the forefront of drug delivery innovation. We offer expert advice to ensure our clients’ strategies effectively meet market demands.
Optimizing Outcomes through Agile Strategies
We remain flexible and responsive to changing requirements by adopting an agile methodology. By employing a risk-based approach, we identify and address potential issues throughout the project.


Capabilities

Positioned for Innovation
Located at the heart of innovation in Cambridge, UK, and Raleigh, North Carolina, our bases are strategically positioned to tap into the world’s leading research and development communities.

ISO-Certified Management System
To support our product development and prototype manufacturing services, CDP’s Quality Management System is certified to ISO 13485:2016 by Intertek Medical Notified Body and ISO 9001:2015 by Intertek Certification Ltd. We are also certified to ISO 27001:2022 by Intertek Certification Ltd to support the maintenance of the confidentiality, availability and integrity of our clients data.

Client-Centric Intellectual Property Approach
Our clients fully own all intellectual property we help create. We equip them with the tools and knowledge to enable lasting independence.
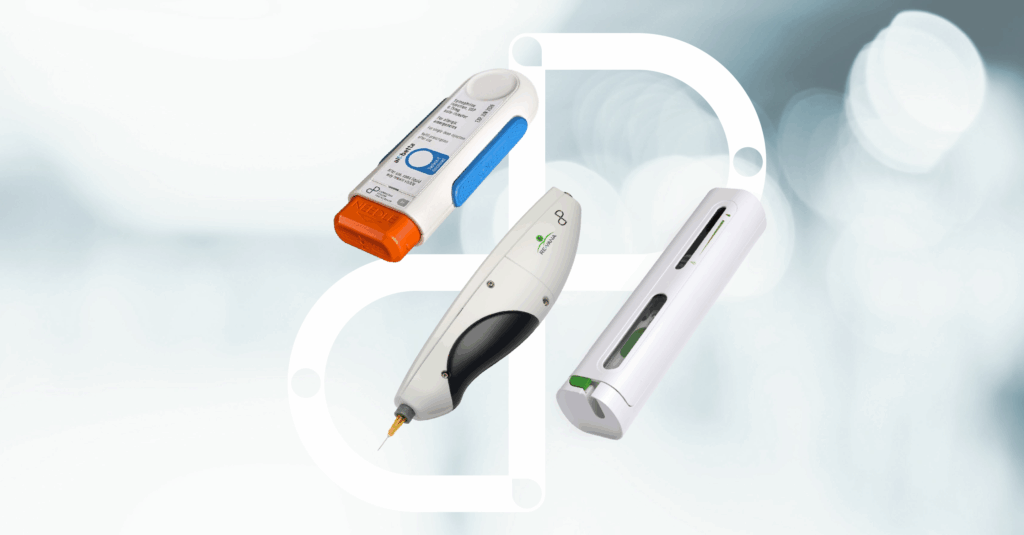
Advancing Injectable Drug Delivery Systems
Unlock expert opinions and insights with four in-depth interviews on the challenges of parenteral innovation.
Breathing New Life: Industry Insights into the future of Respiratory Drug Delivery
Discover the exciting possibilities on the horizon for inhalation device design with our latest resource. This e-book explores recent advancements and highlights innovative strategies crucial for developing significant new treatments.
Meet The Team
Featured Insights

Delivering complexity: device considerations for two-component injectable formulations

Designed to deliver: How collaboration created an award-winning device that puts patients first
Drug Delivery to the Brain: Engineering Precision Across Novel Modalities
FAQs
Explore our FAQs to find out more about how our Drug Delivery team can accelerate your drug delivery device design and enhance user experience.
Who are your typical drug delivery clients?
Mainly a split between BioPharma companies bringing drug products to the market using drug delivery devices and Medtech companies developing and manufacturing drug delivery devices
What typical consultancy services to you provide?
Our project portfolio spans from small, standalone projects to longer projects spanning multiple elements of the development cycle of devices. Services range from very early markets insights, technology assessments, landscaping, early concept generation sprints, to prototyping, Usability Engineering incl. HF studies, Development under full Design Controls up to and including Pilot Manufacturing in-house for e.g. Clinical Trials and Design Transfers to CMO’s
Which types of drug delivery devices do you have experience on?
We have experience on working accross most drug delivery routes of administration, majority within parenteral administration for home uses. Most projects we get involved in often involve complex formulations such as high viscosity, high volume, sensitive API’s. Over the last years, we have seen significant growth in projects within new modalities and targeted delivery, such as intraocular delivery, direct delivery to the brain and nose-to-brain delivery.